
Genes are removed from the virus so it won’t replicate and spread in the body. 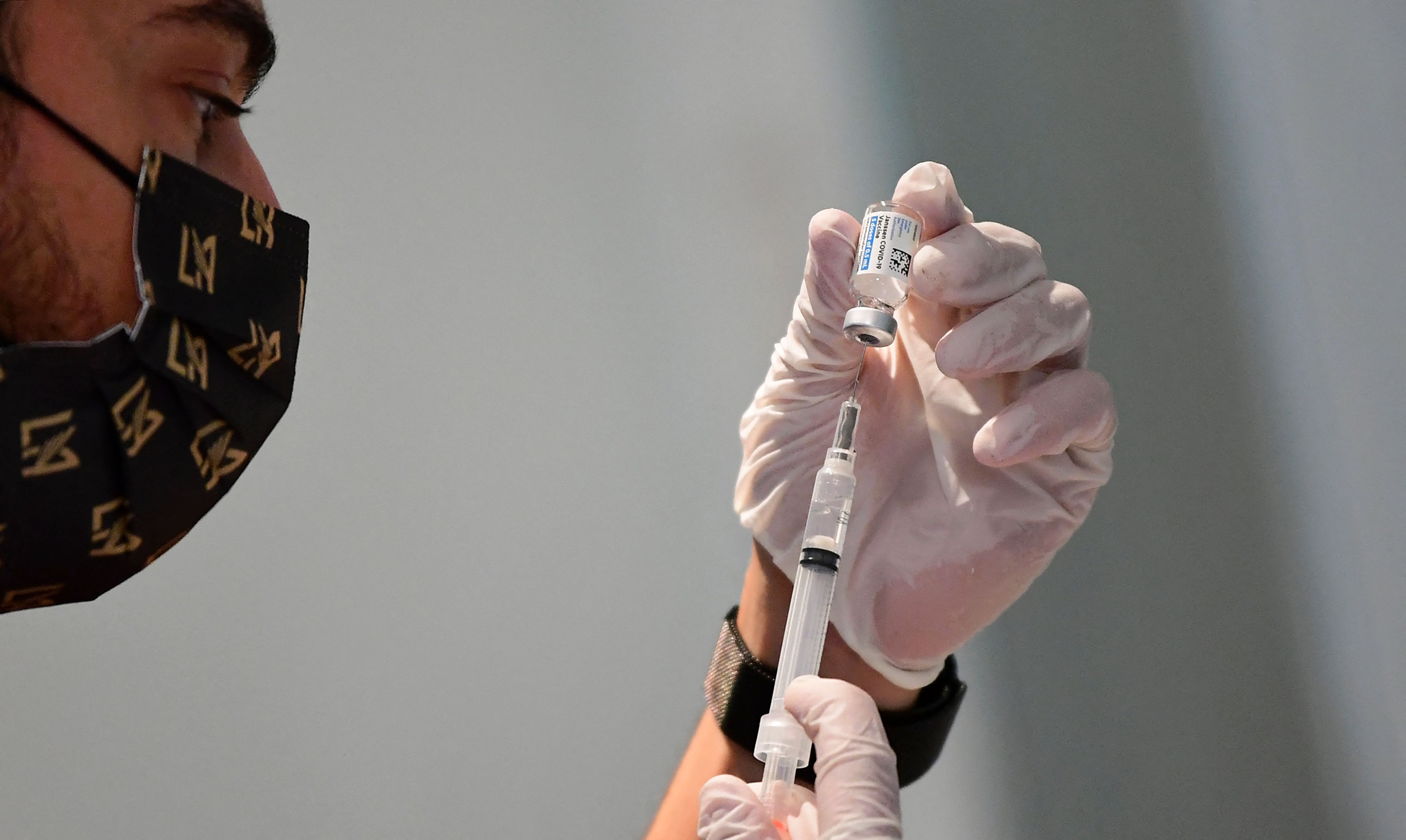
The company that actually starts the manufacturing process for J&J is not Catalent, but Baltimore-based Emergent Biosolutions.Įmergent does the work genetically engineering a virus known as adenovirus 26 that causes the common cold in humans. The plant that, decades ago, made TVs is now helping make the substance that the country is counting on to end its isolation. Everyone at the site feels a great responsibility to do that and we’re working day and night to get it done.” “In such a regulated industry, there are certain steps that you just can’t speed up any faster, but I think we are doing amazing things to get the vaccine out as quickly as we have. “We are going as fast as we can,” Leonard said. The doses going out this week come from elsewhere.Ĭatalent hopes to meet the increasing demands for vaccines to help end the pandemic. It started production of the J&J vaccine at the end of January and should get the regulatory authority to ship from the plant soon. Five shifts of 2,000 employees keep the buzzing production lines going 24 hours a day, seven days a week. Leonard works at the bustling Catalent facility in Bloomington, Indiana, that sits in a nondescript building tucked away by the indoor pickleball court and the Budweiser distributor. 
Here's how it's different from the others The US is getting a third coronavirus vaccine. It expects to have additional capacity by the second quarter of the year and will be made in the US, Europe, Asia and Africa.Ī healthcare worker holds the Johnson & Johnson vaccine against the COVID-19 coronavirus before administaring it to another healthcare worker as South Africa proceeds with its inoculation campaign at the Steve Biko Academic Hospital in Pretoria on February 17, 2021. Three have produced test batches of the vaccine. J&J has looked at 100 potential production sites and found eight that meet its needs so far, Nettles said. Paul Stoffels, chief scientific officer for Johnson & Johnson, told CNN, “you can’t accelerate it by yelling at it.” It takes about five or six weeks to genetically engineer and then grow, purify, and finish the vaccine before it can be bottled and sealed and sent on its way to be distributed to vaccine centers. Richard Nettles, J&J’s vice president of US medical affairs told the subcommittee on Oversight and Investigations for the House Committee on Energy & Commerce last week. “The production of our vaccine is a highly complex process that requires very particular capabilities and experiences,” Dr. Without a global vaccine plan, coronavirus variants could lead to untold number of deaths (AP Photo / Eraldo Peres) Eraldo Peres/AP J&J said it has been working with “urgency” to increase production of the vaccine candidate, but it isn’t easy.Ī healthcare worker applies the second dose of China's Sinovac CoronaVac vaccine during a priority vaccination program for the elderly at Bezerra de Menezes Asylum in Brasilia, Brazil, Thursday, Feb. It can’t happen overnight, but companies say vaccine manufacturing has moved at a speed that has never been seen before in the history of pharmaceutical manufacturing. 
‘You can’t accelerate it by yelling at it’īefore the J&J vaccine can ever get into someone’s arm, it must go through a sophisticated and complicated process that involves several companies. There may still be delays, but Biden said Tuesday the United States would have enough Covid-19 vaccine doses for every adult American by the end of May. The White House said it was utilizing the Defense Production Act to help equip two Merck facilities to manufacture the Johnson & Johnson product, including by bolstering “fill-finish” capacity, when the doses are placed in vials, and by increasing availability of the components of the vaccines. The planned partnership was first reported by The Washington Post. The company has said it expects to make enough doses to vaccinate 20 million people by the end of March, and has a goal to produce 1 billion doses globally by the end of the year.īut as President Joe Biden said Tuesday, “it simply wasn’t coming fast enough.”īiden announced that drugmaker Merck will help manufacture the Johnson & Johnson vaccine – a decision made after it became clear that J&J would fall short of its manufacturing goals. With the US Food and Drug Administration’s authorization last week, Johnson & Johnson said about 4 million doses of its coronavirus vaccine would immediately head to states to help meet enormous demand. (AP Photo/Evan Vucci) Evan Vucci/APīiden now says US will have enough vaccine for every adult by the end of May President Joe Biden speaks about efforts to combat COVID-19, in the State Dining Room of the White House, Tuesday, March 2, 2021, in Washington.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
